
Please re-write this article to improve it to explain concepts before they are used in answers or taken for granted as prior knowledge when they have not first been defined in Unit 1. 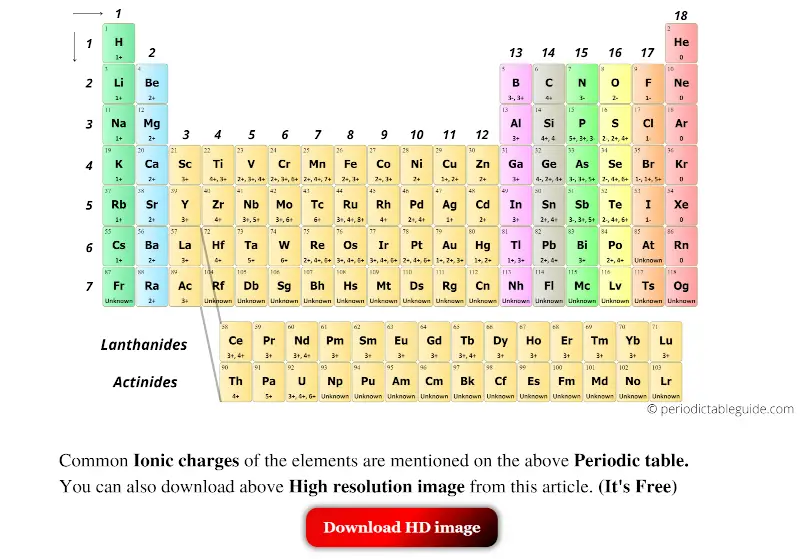
The problems are then for ionic compounds that are not monoatomic. The use of magnitude is used without being introduced followed by a statement that isn't well described, 'The magnitude of the charge is equal to the number of electrons lost, which is equal to the number of valence electrons in the neutral atom.'Īluminum is in group 13, or IIIA is stated when no introduction has been provided to what IIIA means. Where in Unit 1 is s, p, d, f and shell and subshell defined prior to this article. IUPAC group number is also not described, it is simply used, without definition or prior description.Įlectron configuration is used as part of a hidden explanation as an answer, yet the article or Unit 1 does not explain what an electron configuration is. Where in Unit 1 is Octet and Valence given definition. Octet is used without definition, followed by valence also being used without definition. Ions made from alkaline earth metals, the second group on the periodic table, have a 2+ charge. We are being asked to 'Remember that periodic groups refer to columns on the periodic table, whereas rows are known as periods' when this concept has also not been initially defined in this article or in Unit 1. The alkali metals (shown in yellow) always form +1 ions. Where is monatomic defined in this article or in Unit 1. Many of the elements on the periodic table will always form ions that have the same charge. It is important to recognize which numbering system is being used and to be able to find the number of valence electrons in the main block elements regardless of which numbering system is being used.Words are used without first having had a definition or explanation in the article or earlier in Unit 1. You will come across periodic tables with both numbering systems. In this numbering system, group 1A is group 1 group 2A is group 2 the halogens (7A) are group 17 and the noble gases (8A) are group 18. The elements in this group are also gases at room temperature.Īn alternate numbering system numbers all of the \(s\), \(p\), and \(d\) block elements from 1-18. We will learn the reason for this later, when we discuss how compounds form. Review the Periodic Table of the Elements in other formats in Appendix A (credit: Chemistry (OpenStax), CC BY 4.0). Figure 6.1b: Some elements exhibit a regular pattern of ionic charge when they form ions. These elements also have similar properties to each other, the most significant property being that they are extremely unreactive, rarely forming compounds. For example, copper can form ions with a 1+ or 2+ charge, and iron can form ions with a 2+ or 3+ charge. This group contains very reactive nonmetal elements. Group 7A (or 17) elements are also called halogens. It is important to recognize a couple of other important groups on the periodic table by their group name. Question 4 510 Question 7a 165 Question 7b 178. Atoms are electrically neutral because the number of protons, which carry a 1+ charge, in the nucleus of an atom is equal to the number of electrons, which carry a 1- charge, in the atom. 
Image credit: Wikipedia Commons, public domain. Elements from Groups 1 and 17 can combine to form ionic compounds in a one-to-one ratio. Sodium chloride is an ionic compound made up of sodium ions and chloride ions in a crystal lattice. Remember, Mendeleev arranged the table so that elements with the most similar properties were in the same group on the periodic table. Using the periodic table, predict the charge of the most stable ion of the following elements: (e) Sb. Each element in Group 17 can gain one electron to become a 1 anion. The same pattern is true of other groups on the periodic table. Once again, because of their similarities in electron configurations, these elements have similar properties to each other. Group 2A is also called the alkaline earth metals. Although most metals tend to be very hard, these metals are actually soft and can be easily cut. Group 1A is also known as the alkali metals. Because of their similarities in their chemical properties, Mendeleev put these elements into the same group. Ionic charge: When the atom loses or gains one or more electrons, the electric charge is generated (and an ion is formed). The elements in 1A are all very reactive and form compounds in the same ratios with similar properties with other elements. This is what causes these elements to react in the same ways as the other members of the family. All of the 1A elements have one valence electron. A group is a vertical column of the periodic table. Remember that Mendeleev arranged the periodic table so that elements with the most similar properties were placed in the same group.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
